About HOVON
HOVON (Hematology-Oncology for Adults Netherlands) is a non-profit foundation, established in 1985, dedicated to improving the treatment and outcomes of patients with hematological disorders. The HOVON office is located at the Erasmus Medical Center in Rotterdam.
Mission:
"Through national and international collaboration, to optimize the care of patients with hematological diseases by designing and conducting clinical studies and facilitating translational/clinical research through data collection and the establishment of clinically annotated biobanks as well as by other means that further the goal."
Core Tasks and Objective
HOVON conducts clinical, biomarker, and translational studies and develops disease-specific guidelines. The organization connects clinicians and researchers in the Netherlands and Belgium and collaborates with international centers.
Primary Objectives:
- To conduct reliable and independent clinical-translational research supported by Dutch professionals and patient associations.
- To establish study-related databases, biobanks, and innovative research methods.
- To facilitate optimal use of clinical study data and samples to advance knowledge in areas ranging from basic scientific research to quality of life and cost-effectiveness.
- To support registries for disease populations within hematological care and make them available for further research.
- To promote and safeguard knowledge within the hematology field in the Netherlands.
- To ensure a strategically, financially, and operationally sustainable research portfolio.
Organizational Structure
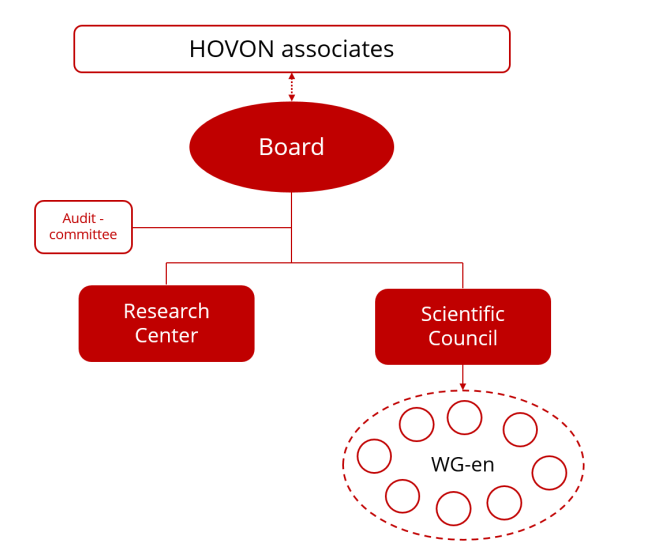
HOVON is organized into six main components:
- Board
Composed of three hematologist board members, a Chief Executive Officer (CEO), and a Chief Financial Officer (CFO).
Collectively responsible for scientific leadership and operational management. - Scientific Council
Includes chairs of disease-specific and modality-specific working groups and representatives from peripheral centers.
Provides strategic and scientific advice to the Board regarding research portfolio alignment and prioritization. - Research Center
Responsible for daily management and execution of clinical research.
Coordinates processes and resources to ensure high-quality research and acts as a liaison to the Board. - HOVON Associates
A meeting of invited participants from academic and peripheral hospitals and other stakeholders.
Enhances involvement in strategy and execution and facilitates feedback to the Board regarding study participation and implementation. - Working Groups
Initiate scientific studies and support HOVON activities.
Three types: disease-specific, treatment modality-specific, and technical support groups. - Audit Committee
Oversees and advises on financial and organizational processes.
Working Groups
Disease-Specific Working Groups:
- Myeloma
- Leukemia (AML, ALL)
- CLL and related disorders
- CML/MPN
- Lymphoma (Hodgkin, Non-Hodgkin)
- LLPC
- Histiocytic-Lymphocytic disorders
- Benign disorders
Treatment Modality-Specific Working Groups:
- IEC (Immune Effector Cell)
- SCT (Stem Cell Transplantation)
- Supportive Care
Technical Working Groups:
- HOP (Pathology Facility and Biobank)
- Imaging
- Molecular Diagnostics
- WSNB (Stem Cell Laboratories Netherlands Belgium)
Guidelines are developed under the auspices of the Nederlandse Vereniging voor Hematologie, with patient association representation.
Governance and Board
- HOVON Board is responsible for strategy, policy, and achieving the foundation’s objectives.
- Composition: minimum of three hematologist board members and two executive board members (CEO & CFO).
- Areas of Focus for Hematologist Board Members:
Scientific Council, study portfolio, statistics, databases, registries, internationalization, stakeholders, and DHC. - Executive Board Members: jointly lead the Research Center.
The HOVONBoard members (2026)
Chair
Dr. N.P.M. Schaap (Radboudumc, Nijmegen)
Vice chair
Prof. Dr. M.H.G.P. Raaijmakers (Erasmus MC, Rotterdam)
Representant of non-academical hospitals
Dr. E.J.F.M. de Kruijf (OLVG, Amsterdam)
CEO
Dr. M.C. Breems- de Ridder
CFO
Drs. B.J.C. ten Bosch
Board Scientific Council (2026)
Chair
Prof. Dr. A.P. Kater (AmsterdamUMC, Amsterdam)
Vice chair
pending
Secretary
pending
