HOVON HO25 NHL
Gearchiveerd
Main info
- Identificatie:
- HO25 NHL / OG 95-004
- Sponsor:
- HOVON
- Stadium:
- 1st lijn
- Included patients:
-
411
- Active sites:
-
0(of 52)52 sites are pending
- Title:
Effect of Recombinant G-CSF on the Results of Chemotherapy (CHOP) in Elderly Patients with Intermediate-/High-Grade Non-Hodgkin's Lymphoma: A Prospective Phase III Study.
Timeline
Scheduled
Actual
1994
01 aug.
Activated
2004
01 aug.
Archived
2015
04 sep.
Closeout in Progress
2030
04 sep.
Destruction
Flow
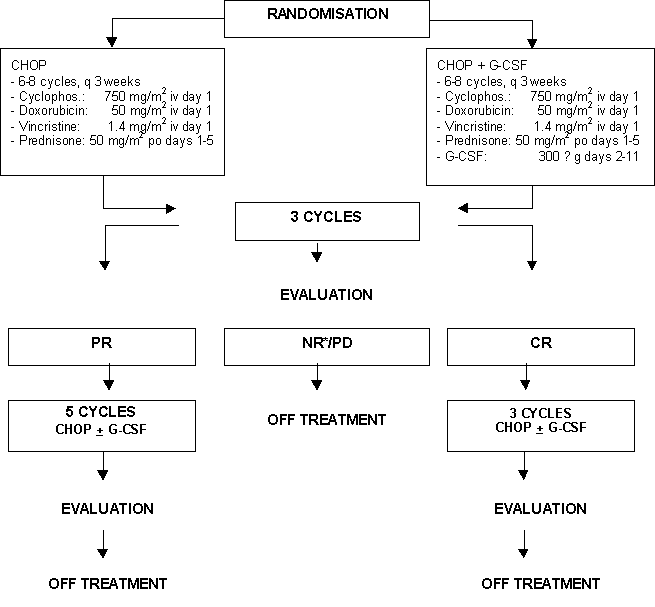
Details
- Phase:
- Prospective randomized Phase III study
- Monitoring Type:
- Not any more
- Objectives:
Eligibility
- Inclusion Criteria:
- Patients with previously untreated Non-Hodgkin's Lymphoma, Ann Arbor stages II-IV, of intermediate- or high-grade malignancy.
- Patients with age 65 years or more.
- Informed consent.
- Exclusion Criteria:
- Patients aged less than 65
- Patients who have been treated for Non-Hodgkin's Lymphoma by chemotherapy or radiotherapy.
- Other malignant diseases, except localized squamous skin carcinoma.
- Severe heart failure.
- Inadequate liver or renal function.
- Failure to give informed consent.
- HIV positivity.
Registration Details
Registration
Central registration at the HOVON Data Center:
Erasmus MC Cancer Institute, Clinical Trial Center (Hs-423)
P.O. Box 2040
NL-3000 CA Rotterdam
Phone number.: +31.10.7041560 (working days 9.00-17.00)
Fax number.....: +31.10.7041028
TOP address...: http://www.hdc.hovon.nl/top
Registration criteria
The following information will be requested:
- Name institute
- Name of caller and/or physician in charge
- Hospital record number
- Patient name, date of birth and sex
- Pathology diagnosis
- Stage II, III or IV, A/B
- Serum LDH
Participating Sites
Site
52 results
Order by
Accrual rate
Activation date
NL-Rotterdam-ERASMUSMC
29
NL-Amsterdam-VUMC
27
NL-Amersfoort-MEANDERMC
22
NL-Zaandam-ZAANSMC
22
BE-Leuven-UZLEUVEN
21
NL-Enschede-MST
19
NL-Den Haag-HAGA
18
NL-Utrecht-UMCUTRECHT
16
NL-Groningen-UMCG
12
NL-Amsterdam-AMC
12
NL-Rotterdam-EMCDANIEL
12
NL-Haarlem-SPAARNEHAARLEM
11
NL-Leeuwarden-MCL
11
NL-Zwolle-ISALA
11
NL-Leiden-LUMC
11
NL-Amsterdam-OLVG
11
NL-Rotterdam-MAASSTADZIEKENHUIS
10
NL-Eindhoven-CATHARINA
10
NL-Nieuwegein-ANTONIUS
9
NL-Schiedam-FRANCISCUSVLIETLAND
8
NL-Roosendaal-BRAVIS
8
NL-Hilversum-TERGOOI
7
NL-Leiderdorp-ALRIJNELEIDERDORP
7
NL-Dordrecht-ASZ
6
NL-Eindhoven-MAXIMAMC
6
NL-Breda-AMPHIA
6
NL-Amsterdam-AVL
5
NL-Amsterdam-SLOTERVAART
5
NL-Tilburg-ETZ
5
NL-Vlaardingen-VLIETLANDVLARDINGEN
5
NL-Utrecht-DIAKONESSENUTRECHT
4
NL-Winterswijk-SKBWINTERSWIJK
4
NL-Leidschendam-HMCANTONIUSHOVE
4
NL-Purmerend-DIJKLANDERPURMEREND
3
NL-Hoorn-DIJKLANDERHOORN
3
NL-Veghel-BERNHOVENVEGEL
3
NL-Dirksland-VANWEELBETHESDA
3
NL-Den Bosch-JBZ
2
NL-Delft-RDGG
2
NL-Groningen-MARTINI
2
NL-Tilburg-TWEESTEDEN
2
NL-Geldrop-STANNA
2
NL-Apeldoorn-GELREAPELDOORN
2
NL-Stadskanaal-TRENTREFAJA
2
NL-Emmen-SCHEPER
2
NL-Meppel-ISALADIACONESSEN
1
NL-Heerenveen-TJONGERSCHANS
1
NL-Gorinchem-BEATRIX
1
NL-Capelle a/d IJssel-YSL
1
NL-Sittard-Geleen-ZUYDERLAND
1
NL-Gouda-GROENEHART
1
NL-Den Helder-NWZDENHELDER
1
