HOVON HO29 AML
Gearchiveerd
Main info
- Identificatie:
- HO29 AML / SAKK 30/95
- Sponsor:
- HOVON
- Included patients:
-
1105
- Active sites:
-
0(of 35)35 sites are pending
- Title:
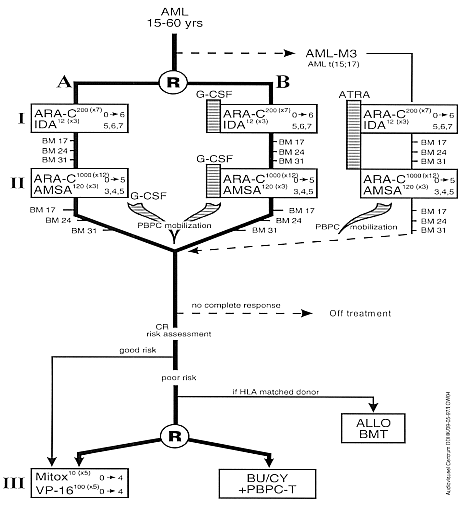
Remission Induction Chemotherapy with or without rHuG-CSF (lenograstim) sensitization.
Marrow Ablative Chemotherapy plus Autologous Peripheral Progenitor/Stem Cell Transplantation (PBSCT) versus Chemotherapy.
Timeline
Scheduled
Actual
1995
15 mrt.
Activated
1998
01 dec.
ClosedForInclusionActualStart
2015
13 okt.
Archived
2030
13 okt.
Destruction
Flow
Details
- Phase:
- Prospective randomized Phase III study
- Monitoring Type:
- Objectives:
Eligibility
- Inclusion Criteria:
- Patients with newly diagnosed de novo AML.
- Age 15-60 years inclusive.
- Patients have given infomed consent.
- Leucocytosis is not an exclusion criterium, but it will require postponement of G-CSF administration until WBC have declined to 20 x 109/l on chemotherapy.
- Exclusion Criteria:
A concurrent active malignancy, except stage I cervix carcinoma and basocellular carcinoma.
- Previous treatment with chemotherapy.
- Leukemia following from a documented myelodysplasia with a duration of more than 6 months.
- Blastic crisis of chronic myeloid leukemia or leukemia developing from myeloproliferative diseases.
- Renal or liver function abnormalities.
- HIV positive serology.
- Severe cardiac, pulmonary or neurologic disease.
Participating Sites
Site
35 results
Order by
Accrual rate
Activation date
NL-Amsterdam-VUMC
94
BE-Leuven-UZLEUVEN
86
NL-Groningen-UMCG
85
CH-Zürich-USZ
73
NL-Utrecht-UMCUTRECHT
67
NL-Rotterdam-ERASMUSMC
65
NL-Rotterdam-EMCDANIEL
63
CH-Bern-INSEL
42
NL-Maastricht-MUMC
41
BE-Bruxelles-STLUC
39
CH-Basel-USB
32
NL-Amsterdam-AMC
30
CH-Lausanne-CHUV
30
NL-Den Haag-HAGA
29
NL-Enschede-MST
26
BE-Yvoir-MONTGODINNE
25
CH-St. Gallen-KSSG
24
CH-Aarau-KSA
23
NL-Zwolle-ISALA
22
CH-Geneve (14)-HCUGE
20
NL-Amersfoort-MEANDERMC
19
NL-Nieuwegein-ANTONIUS
13
NL-Amsterdam-AVL
12
BE-Haine-Saint-Paul-JOLIMONT
10
CH-Neuchatel-HNE
10
CH-Bellinzona-SANGIOVANNI
9
CH-Winterthur-KSW
7
DE-Frankfurt am Main-NORDWEST
4
NL-Meppel-ISALADIACONESSEN
4
NL-Delft-RDGG
2
NL-Hilversum-TERGOOI
1
NL-Leeuwarden-MCL
1
NL-Amsterdam-SLOTERVAART
1
NL-Eindhoven-CATHARINA
1
DE-Mainz-UNIMEDIZINMAINZ
