HOVON HO35 NHL
Gearchiveerd
Main info
- Identificatie:
- HO35 NHL / EORTC 20963
- Sponsor:
- HOVON
- Included patients:
-
65
- Active sites:
-
0(of 27)27 sites are pending
- Title:
Marrow-Ablative Chemo-Radiotherapy and Autologous Stem Cell Transplantation followed by Interferon-Alpha Maintenance Treatment versus Interferon-Alpha Maintenance Treatment alone after a Chemotherapy-Induced Remission in Patients with Stages III or IV Follicular Non-Hodgkin's Lymphoma: A Prospective, Randomized, Phase III Clinical Trial - Intergroup collaborative study (EORTC 20963).
Timeline
Scheduled
Actual
1997
27 feb.
Activated
1999
05 apr.
ClosedForInclusionActualStart
2009
05 apr.
Archived
2034
05 apr.
Destruction
Flow
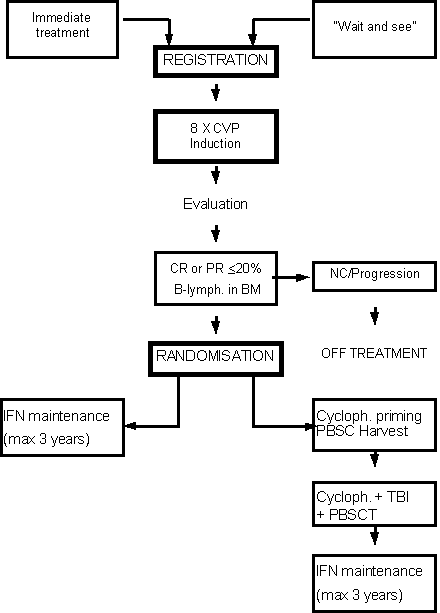
Details
- Phase:
- Prospective randomized Phase III study
- Monitoring Type:
- Objectives:
Eligibility
- Inclusion Criteria:
- Previously untreated patients with follicular NHL presenting with stages III or IV, including nodal, Waldeye's ring and extranodal localisations.
- Follicular NHL according to the: - REAL classification (follicle center lymphoma, follicular provisional cytologic grades: I, II, III) - International Working Formulation (Groups B, C, D) - Kiel Classification (malignant lymphoma)
- Age above 18 years and below 66 years.
- WHO performance Status grade 0, 1 or 2.
- Patient information and informed consent according to the rules of the respective country.
- Exclusion Criteria:
- Patients with severe cardiac, pulmonary, neurologic, psychiatric or metabolic disease.
- Serum creatinine, BUN, alkaline phoshatase or bilirubin: = 2.5 times the upper limit of the normal value, unless clearly related to NHL.
- Pregnant women.
- Patients with prior malignancies except non-melanoma skin tumors or stage 0 cervical carcinoma.
- Patients with HIV positivity.
- Patients who are unable to attend regular outpatient appointments for treatment and treatment evaluation.
Participating Sites
Site
27 results
Order by
Accrual rate
Activation date
NL-Rotterdam-ERASMUSMC
8
NL-Nijmegen-RADBOUDUMC
8
NL-Amersfoort-MEANDERMC
7
NL-Rotterdam-EMCDANIEL
5
BE-Leuven-UZLEUVEN
4
NL-Zwolle-ISALA
4
NL-Enschede-MST
4
NL-Groningen-UMCG
3
NL-Roosendaal-BRAVIS
2
NL-Amsterdam-AMC
2
NL-Delft-RDGG
2
NL-Spijkenisse-SPIJKENISSEMC
1
NL-Rotterdam-IKAZIA
1
NL-Sittard-Geleen-ZUYDERLAND
1
NL-Schiedam-FRANCISCUSVLIETLAND
1
NL-Heerlen-ATRIUMMC
1
NL-Tilburg-TWEESTEDEN
1
NL-Eindhoven-MAXIMAMC
1
NL-Goes-ADRZ
1
NL-Amsterdam-SLAZLUCAS
1
NL-Utrecht-UMCUTRECHT
1
NL-Rotterdam-MAASSTADZIEKENHUIS
1
NL-Den Bosch-JBZ
1
NL-Dirksland-VANWEELBETHESDA
1
NL-Nieuwegein-ANTONIUS
1
NL-Hilversum-TERGOOI
1
NL-Vlaardingen-VLIETLANDVLARDINGEN
1
