HOVON HO78
Gearchiveerd
Main info
- Identificatie:
- HOVON 78 CML
- Sponsor:
- HOVON
- Included patients:
-
111
- Active sites:
-
29(of 29)
- Title:
Imatinib in combination with Cytarabin as compared to Imatinib alone in patients with first chronic phase Chronic Myeloid Leukemia. A prospective randomized study.
Timeline
Scheduled
Actual
2006
24 mei
Activated
2010
01 mei
Closeout in Progress
2015
13 okt.
Archived
2030
13 okt.
Destruction
Flow
Details
- Phase:
- Prospective randomized Phase III study
- Monitoring Type:
- Objectives:
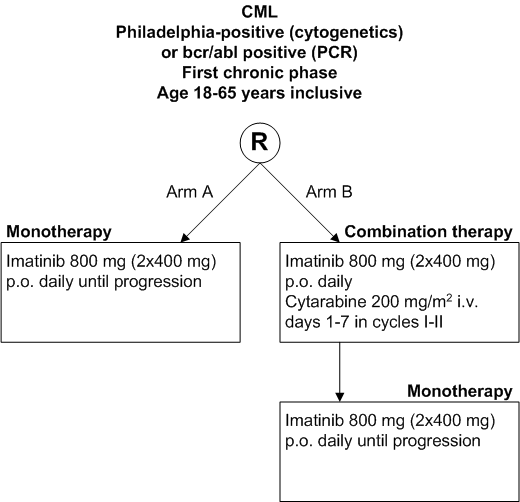
To determine the efficacy of the combination of imatinib with cytarabine as compared to imatinib alone in terms of the rate of molecular response at 12 months from randomization.
Eligibility
- Inclusion Criteria:
- Newly diagnosed patients with CML in first chronic phase ? 2 months;
- Presence of Philadelphia chromosome or bcr-abl rearrangement;
- Age 18-65 years inclusive;
- WHO performance status ? 2 (see appendix E);
- Written informed consent.
- Exclusion Criteria:
- CML in accelerated phase or blastic crisis as defined by the WHO criteria (see appendix A).
- Hepatic dysfunction (serum bilirubin ? 2 x N, and/or ALAT ? 4 x N, and/or ASAT ? 4 x N);
- Renal dysfunction (creatinine ? 200 ?mol/l or 2.3 mg/dl);
- Severe cardiac dysfunction (NYHA classification II-IV, see appendix F);
- Severe pulmonary or neurologic disease;
- Pregnant or lactating females;
- Patients with a history of active malignancy during the past 5 years with the exception of
basal carcinoma of the skin or stage 0 cervical carcinoma;
- Patients known to be HIV-positive;
- Patients with active, uncontrolled infections;
- Previous treatment other than hydroxyurea ? 2 months or imatinib ? 1 month;
- Male and female patients of reproductive potential who are not practicing effective means of
contraception.
Participating Sites
Site
29 results
Order by
Accrual rate
Activation date
NL-Rotterdam-EMCDANIEL
17
NL-Rotterdam-ERASMUSMC
15
NL-Amsterdam-VUMC
13
NL-Amsterdam-AMC
8
NL-Leiden-LUMC
8
NL-Den Haag-HAGA
6
NL-Den Bosch-JBZ
6
NL-Enschede-MST
5
NL-Zwolle-ISALA
5
NL-Nijmegen-RADBOUDUMC
5
NL-Dordrecht-ASZ
4
BE-Leuven-UZLEUVEN
3
NL-Amersfoort-MEANDERMC
3
NL-Hoofddorp-SPAARNEGASTHUIS
2
NL-Utrecht-UMCUTRECHT
2
NL-Nieuwegein-ANTONIUS
2
NL-Maastricht-MUMC
2
NL-Breda-AMPHIA
2
NL-Hilversum-TERGOOI
1
NL-Groningen-UMCG
1
BE-Antwerpen-ZNASTUIVENBERG
1
NL-Schiedam-FRANCISCUSVLIETLAND
NL-Amsterdam-SLOTERVAART
NL-Heerlen-ATRIUMMC
BE-Antwerpen-ZNAMIDDELHEIM
NL-Delft-RDGG
NL-Hoorn-DIJKLANDERHOORN
NL-Roosendaal-BRAVIS
NL-Sittard-Geleen-ZUYDERLAND
